Valproic Acid and Mitochondria
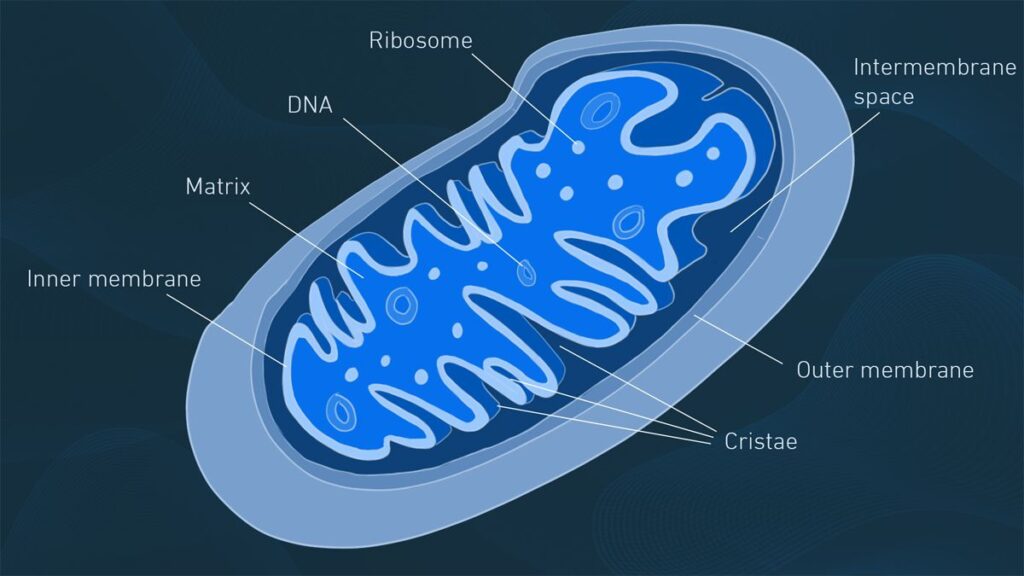
Valproic acid (VPA) is known to induce mitochondrial dysfunction, primarily through its interference with mitochondrial beta-oxidation, which can lead to liver toxicity and steatosis. This effect is attributed to VPA’s structural resemblance to fatty acids, allowing it to enter mitochondria and generate a coenzyme A ester (VPA-CoA) that inhibits key enzymes involved in fatty acid oxidation, such as carnitine palmitoyltransferase-1 (CPT1). Additionally, VPA can inhibit α-lipoamide dehydrogenase, an enzyme critical for pyruvate oxidation, further disrupting mitochondrial function. Prolonged exposure to VPA has been shown to reduce mitochondrial respiration and alter gene expression, particularly involving transcription factors like C/EBPα, which plays a role in mediating mitochondrial dysfunction.
- Mechanism of Mitochondrial Dysfunction: Valproic acid (VPA) interferes with mitochondrial beta-oxidation by generating VPA-CoA, which inhibits CPT1 and reduces mitochondrial levels of CoA, a cofactor essential for fatty acid oxidation. This inhibition leads to impaired energy production and lipid accumulation in hepatocytes.
- Liver Toxicity: VPA is associated with liver toxicity, which is often linked to mitochondrial dysfunction. The drug can cause steatosis and necrosis by disrupting mitochondrial β-oxidation and reducing antioxidant capacity.
- Impact on Gene Expression: Prolonged exposure to VPA can alter gene expression, particularly affecting pathways related to mitochondrial function. The transcription factor C/EBPα has been identified as a key player in VPA-induced mitochondrial dysfunction, with its knock-down partially rescuing the adverse effects.
- Inhibition of α-Lipoamide Dehydrogenase: VPA strongly inhibits α-lipoamide dehydrogenase, an enzyme involved in the pyruvate dehydrogenase complex, which is crucial for energy metabolism. This inhibition is more pronounced in liver mitochondria compared to brain mitochondria.
- Case Studies: VPA has been implicated in exacerbating underlying mitochondrial cytopathies, such as mitochondrial myopathy, encephalopathy, lactic acidosis, and stroke-like episodes (MELAS). A case study reported a young boy whose underlying mitochondrial disorder was unmasked by VPA use, leading to severe encephalopathy and hepatic damage.
Valproic Acid’s Mitochondrial Toxicity and Clinical Safety Implications
Valproic acid (VPA) exerts several negative effects on mitochondrial function through multiple mechanisms, which has significant clinical safety implications for bipolar patients maintained on this medication.
Mechanisms of Mitochondrial Toxicity
1. Inhibition of Key Enzymes The primary mechanism involves VPA’s inhibition of α-lipoamide dehydrogenase, a crucial enzyme in mitochondrial energy metabolism. This severely impairs pyruvate and 2-oxoglutarate oxidation in liver mitochondria at concentrations above 100 μM, which are within the therapeutic range.
2. Disruption of β-Oxidation VPA causes disruption of mitochondrial β-oxidation, which is essential for lipid metabolism. This disruption leads to microvesicular hepatosteatosis, a characteristic feature of VPA toxicity that suggests direct mitochondrial involvement.
3. Impaired Mitochondrial Bioenergetics VPA treatment decreases mitochondrial membrane potential and oxygen consumption, forcing cells to compensate by increasing glycolysis. This metabolic shift indicates significant impairment of normal mitochondrial energy production.
4. Compromised Mitochondrial Biogenesis While VPA triggers increased mitochondrial biogenesis, cells with pre-existing mitochondrial dysfunction (such as POLG-deficient cells) cannot adequately address the increased metabolic demands, leading to cellular stress and toxicity.
Clinical Safety Implications for Bipolar Patients
1. Hepatotoxicity Risk VPA is a well-known cause of several distinctive forms of acute and chronic liver injury. The hepatotoxicity is believed to be mediated by VPA’s inhibitory effect on the mitochondrial β-oxidation pathway and VPA-induced metabolic effects.
2. Particularly High-Risk Populations Patients with mitochondrial diseases, specifically POLG-related mitochondrial disorders, have amplified risk of hepatotoxicity and mortality when given VPA. The medication should only be used in patients older than 2 with suspected mitochondrial disorders who have not responded to other treatments.
3. Age-Related Vulnerability Hepatotoxicity is particularly severe in those younger than 2 years old and in patients on polytherapy, reflecting the increased vulnerability of developing mitochondrial systems and the additive stress of multiple medications.
4. Oxidative Stress and Antioxidant Depletion VPA reduces antioxidant capacity and promotes necrosis, factors that contribute to hepatic steatosis and liver damage. This creates a cascade of cellular damage that can be particularly problematic in long-term treatment.
Clinical Monitoring Recommendations
Given these mitochondrial effects, bipolar patients on valproate require:
- Regular liver function monitoring, especially during the first few months of treatment
- Careful screening for mitochondrial disorders before initiating therapy
- Monitoring for signs of metabolic dysfunction, including weight gain and metabolic syndrome
- Assessment of risk factors such as age, polytherapy, and underlying metabolic conditions
- Patient and family education about early signs of hepatotoxicity
Risk-Benefit Considerations
While VPA remains an effective mood stabilizer for bipolar disorder, its mitochondrial toxicity necessitates careful patient selection and monitoring. The mechanism involves oxidative stress and genetic factors, with microvesicular hepatosteatosis being a typical feature suggestive of mitochondrial β-oxidation involvement.
Clinicians must weigh the therapeutic benefits against the potential for serious mitochondrial-mediated toxicity, particularly in vulnerable populations. Alternative mood stabilizers should be considered for patients with known or suspected mitochondrial dysfunction, very young patients, or those with multiple risk factors for hepatotoxicity.
The mitochondrial effects of VPA represent a classic example of how drug efficacy and toxicity can share common mechanistic pathways, emphasizing the importance of personalized medicine approaches in psychiatric treatment.
Evidence for Valproic Acid’s Mitochondrial Toxicity: Both Human Clinical Evidence and Laboratory Studies
The negative effects of valproic acid on mitochondria are supported by both human clinical evidence and experimental laboratory studies. Here’s the breakdown:
Human Clinical Evidence
1. Clinical Observations in Patients Clinical observations have documented that liver toxicity of VPA is extremely problematic in Alpers-Hüttenlocher syndrome—a mitochondrial form of epilepsy due to mutations in the mitochondrial DNA polymerase γ affecting brain and liver. This represents direct human clinical evidence.
2. Systematic Clinical Analysis A systematic analysis of VPA toxicity in mitochondrial diseases found fatal outcomes in 53/102 (52%) POLG VPA-exposed patients, while there were no fatal adverse drug reactions in the mtDNA disease group (35 patients), and only 1 out of 54 patients with non-POLG mitochondrial disease developed acute liver failure. This demonstrates clear clinical evidence in humans with mitochondrial disorders.
3. Primary Human Hepatocyte Studies Studies using primary human hepatocytes (PHHs) showed that prolonged VPA exposure (24-72 hours) significantly reduced basal and maximal oxygen consumption rates, while short exposure (3 hours) did not affect these rates. This study illustrated that prolonged VPA exposure promotes mitochondrial dysfunction in human hepatocytes, with mitochondrial dysfunction appearing to be driven by C/EBPα-dependent gene expression.
4. Clinical Case Reports Case reports document VPA exacerbating underlying mitochondrial cytopathy, with the primary mechanism being interference with mitochondrial beta-oxidation. Clinical cases of VPA-induced acute liver injury resulting in hepatic encephalopathy have been documented, with hyperammonemia being a common complication affecting 27.8% of patients.
Laboratory/Experimental Evidence
1. Mechanistic Studies Laboratory studies in rat models showed that VPA concentrations above 100 μM severely impaired liver mitochondrial pyruvate and 2-oxoglutarate oxidation, while brain mitochondria were nearly unaffected.
2. Metabolic Studies Laboratory studies demonstrated that clinically relevant concentrations of VPA increase carbon accumulation through glycolysis and decrease mitochondrial bioenergetics, with VPA directly inhibiting mammalian COX activity.
3. Fibroblast Studies Experimental evidence showed that VPA triggers increased mitochondrial biogenesis by altering the expression of several mitochondrial genes; however, the capacity of POLG-deficient liver cells to address the increased metabolic rate caused by VPA administration is significantly impaired.
Clinical Relevance
The liver toxicity of VPA is an established side effect of this widely used antiepileptic drug, which is extremely problematic for patients with metabolic epilepsy and particularly epilepsy due to mitochondrial dysfunction. VPA is known to cause liver toxicity and steatosis through mitochondrial dysfunction in clinical practice.
Conclusion
The evidence for valproic acid’s negative effects on mitochondria is robust and multifaceted, combining:
- Direct clinical observations in patients with mitochondrial diseases
- Systematic clinical analyses showing increased mortality in vulnerable populations
- Studies using primary human hepatocytes demonstrating mitochondrial dysfunction
- Case reports documenting clinical manifestations
- Laboratory studies elucidating the underlying mechanisms
This combination of human clinical evidence and experimental data provides a strong foundation for understanding VPA’s mitochondrial toxicity as a clinically relevant phenomenon, not just a laboratory finding. The clinical evidence is particularly compelling in patients with underlying mitochondrial disorders, where VPA toxicity can be fatal.